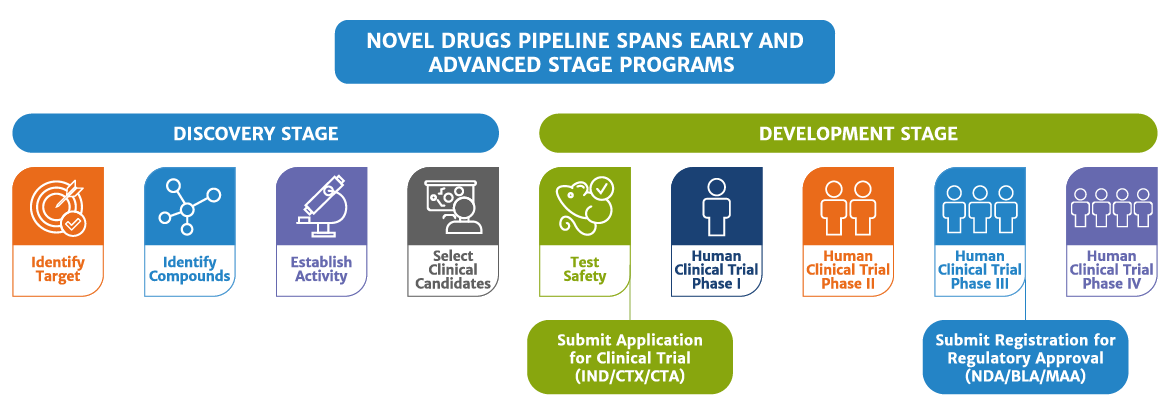
Our novel assets under development combine early and advanced stage programs. The foray into novel biologics predates our entry into the biosimilars segment. We are the pioneers in developing, manufacturing and launching BIOMAb-EGFR®, India’s first indigenously produced novel monoclonal antibody for the treatment of head and neck cancer; and ALZUMAb™, the world’s first novel anti-CD6 monoclonal antibody in India, for psoriasis.

BIOCON AND NOVELS
Biocon & Novels
Addressing unmet needs in cancer and autoimmune conditions

The foray into novel biologics predates our entry into the biosimilars segment.

Novels Portfolio
Our current novels portfolio acquired through early stage partnerships include:
Monoclonal antibodies against novel targets like CD6, against established targets like EGFR
A pipeline of bispecific fusion antibodies that exploit the recent understanding of the role of checkpoint inhibitors
Novels Biologics
A rigorous valuation of their ‘functional activities’ and ‘mechanism of action’ helps us map the progress of our programs. Our skills in manufacturing processes, analytical development and cutting-edge translational and clinical sciences enable us to maximise the overall probability of success.
Know more
about us